| Standard No. |
Title |
Price(USD) |
Delivery |
Status |
Add To Cart |
| GB/T 27821-2011 |
Chemicals—Bacterial DNA damage or repair test |
 (USD) (USD) |
via email in
1-3 business day
|
VALID
|
|
| YY/T 1578-2018 |
Glycated albumin assay kit(enzymatic method) |
 (USD) (USD) |
via email in
1-3 business day
|
VALID
|
|
| YY/T 1483-2016 |
Anti-herpes-simplex-virus IgM antibody detection reageant(kit) |
 (USD) (USD) |
via email in
1-3 business day
|
VALID
|
|
| YY/T 1252-2015 |
Total IgE quantitative labelling immunoassay kit |
 (USD) (USD) |
via email in
1-3 business day
|
VALID
|
|
| YY/T 1254-2015 |
High density lipoprotein(HDL)cholesterol assay kit |
 (USD) (USD) |
via email in
1-3 business day
|
VALID
|
|
| YY/T 1235-2014 |
Rubella IgG/IgM antibody detection kit |
 (USD) (USD) |
via email in
1-3 business day
|
VALID
|
|
| GB/T 16886.20-2015 |
Biological evaluation of medical devices—Part 20:Principles and methods for immunotoxicology testing of medical devices |
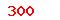 (USD) (USD) |
via email in
1-3 business day
|
VALID
|
|
| YY/T 1197-2013 |
Alanine aminotransferase diagnostic kit (IFCC method) |
 (USD) (USD) |
via email in
1-3 business day
|
VALID
|
|
| GB/Z 30154-2013 |
Medical laboratories—Guidance on laboratory implementation of GB/T 22576—2008 |
 (USD) (USD) |
via email in
1-3 business day
|
VALID
|
|
| YY/T 0456.1-2014 |
Reagents for hematology analyzer—Part 1:Rinse |
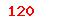 (USD) (USD) |
via email in
1-3 business day
|
|
|
| YY/T 0700-2008 |
Clothing for protection against contact with blood and body fluids—Determination of the resistance of protective clothing materials to penetration by blood and body fluids—Test method using synthetic blood |
 (USD) (USD) |
via email in
1-3 business day
|
VALID
|
|
| GB/T 21415-2008 |
In vitro diagnostic medical devices - Measurement of quantities in biological samples - Metrological traceability of values assigned to calibrators and control materials |
 (USD) (USD) |
via email in
1-3 business day
|
VALID
|
|
| GB/T 35522-2017 |
Chemicals—Collembolan reproduction test in soil |
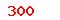 (USD) (USD) |
via email in
1-3 business day
|
VALID
|
|
| YY/T 1259-2015 |
Diagnostic kit for IgG antibody to hepatitis E virus(ELISA) |
 (USD) (USD) |
via email in
1-3 business day
|
VALID
|
|
| YY/T 1233-2014 |
Cardiac troponin-I(cTnI)quantitative detection reagent(kit)(Chemiluminescent immunoassay) |
 (USD) (USD) |
via email in
1-3 business day
|
VALID
|
|
| GB/T 16886.3-2008 |
Biological evaluation of medical devices - Part 3: Tests for genotoxicity carcinogenicity and reproductive toxicity |
 (USD) (USD) |
via email in
1-3 business day
|
ABOLISHED
|
|
| YY/T 1209-2013 |
BCYE agar medium |
 (USD) (USD) |
via email in
1-3 business day
|
VALID
|
|
| YY/T 0501-2004 |
General technical requirements for urinalysis control |
 (USD) (USD) |
via email in
1-3 business day
|
ABOLISHED
|
|
| GB/T 21793-2008 |
Chemicals - Test method of in vitro mammalian cell gene mutation |
 (USD) (USD) |
via email in
1-3 business day
|
VALID
|
|
| YY/T 0576-2005 |
Columbia Blood Agar Base Medium |
|
via email in
1-3 business day
|
VALID
|
please email coc@codeofchina.com for quotation
|
|
|

 (USD)
(USD)  (USD)
(USD)  (USD)
(USD)  (USD)
(USD)  (USD)
(USD)  (USD)
(USD)  (USD)
(USD)  (USD)
(USD)  (USD)
(USD) 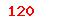 (USD)
(USD)  (USD)
(USD)  (USD)
(USD) 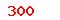 (USD)
(USD)  (USD)
(USD)  (USD)
(USD)  (USD)
(USD)  (USD)
(USD)  (USD)
(USD)  (USD)
(USD)